Fruitless remains ever-fruitful: a genetic story of love and aggression
This article is being cross-listed on The Berkeley Science Review. Check out some other really interesting pieces there!
One of the most exciting prospects of biological inquiry lies in its potential to explain the peculiarities of our own lives. Human behavior provides some of the most spectacular examples of the output of a fundamentally biological system, the brain, but despite decades of remarkable research spanning the scale of neurotransmitters, to brain hemispheres, to interactions on the internet, we struggle to firmly explain the biological (and environmental) underpinnings of any given human behavior.
Luckily, some of our behaviors aren’t exclusive to our species. Chimpanzees seem to mourn the death of family members. Ants are capable of organizing into social hierarchies that eerily resemble human social structures. Even the lowly fruit fly, Drosophila melanogaster, goes through bouts of light and heavy sleep that resemble human sleep cycles. All these examples suggest that the biology of behavior might be conserved in the same way that many genes are conserved from fly to human, and hint that perhaps the same genes control similar behaviors in wildly different organisms – a hint that tantalizes scientists to this day.

Source: Wikipedia
A century ago, when the fruit fly was popularized as a model organism by the lab of Thomas Morgan, these similarities were fairly well-appreciated, but tools for manipulating fly genetics and observing fly behavior were lacking. Fifty years later, researchers were better-prepared to manipulate the fruit fly genome, thanks to advances made in prior decades with mutating the fly genome with x-rays. In 1963, a researcher at Yale named Kulbir Gill used this technology to create mutant flies that lacked the typical behavioral program ensuring male pursuit of female partners for reproduction. These mutant male flies courted male and female partners equally, and utterly failed to reproduce. Gill dubbed this mutation ‘fruity,’ a less-than-appropriate pun on the fruit fly and its mating strategy, but limited his observations to a short note in a fly journal, declining to investigate further.
In 1977, the geneticist Jeffrey Hall dug up Gill’s fruity line, and again observed a failure of male flies to properly court and reproduce with female flies. Hall renamed the mutation fruitless, acknowledging Gill’s original name while tactfully shifting its implication onto these flies’ futile attempts at reproduction. With Hall’s resurrection of the fruitless fly line, scientists now had an animal model that could provide insight into how genes influence male behavior – an insight with the potential to explain the root of differences in the behavior of men and women.
We now know that the original fruitless flies possessed a mutation in the fruitless gene, which disrupts the production of the male-specific protein FruM. FruM is expressed in neurons that exist in the male, but not female, fly brain, and some of these neurons are responsible for orchestrating the male courtship ritual. When scientists engineered female flies to express FruM, these female flies began to behave like courting males, despite their lack of the very neurons that guide male courtship. All this raises some of the biggest questions in biology. If sexual orientation and courtship can be influenced by a single gene in flies, might these traits be controlled by a gene, or genes, in humans? If differential gene expression in female versus male flies leads to behavioral differences, does a similar scenario explain behavioral differences across the human sexes? And lastly, how does the introduction of a male protein into a female brain allow that female brain to carry out a male behavior, when the male neural circuitry is not present?
FruM, and the male neurons possessing FruM, obviously deserved further study, as biological mediators of male behavior. Over the course of his career, Hall himself managed to define the FruM neurons responsible for male courtship, but these neurons represented but a fraction of the total of two thousand FruM neurons, suggesting that FruM might control other male-specific behaviors in the fly. High on the list of observable sexually-dimorphic behaviors in the fly is aggression: though both male and female flies are capable of exhibiting aggression, males are measurably more aggressive than females. This is also true for most mammalian species, including humans. Men are broadly more violent than women, and while the causes of human violence are likely more complex than any violence observed in a fly vial, it is tempting to speculate on how biology, and genetics, might be influencing this behavioral sexual dimorphism, just like how FruM neurons enable male flies to perform their courtship song.
Perhaps unsurprisingly, aggression was formally linked to fruitless by the lab of Barry Dickson at Harvard in 2006, with a study that was able to swap male and female modes of aggressive fighting by replacing the fruitless variant of one gender with the fruitless variant from the opposite gender. However, despite the efforts of dozens of additional labs, the identity of the FruM neurons that drove male-specific aggression remained unknown. Furthermore, even though both courtship and aggression in flies seemed to depend on fruitless, this gene was nowhere to be found in higher organisms, including humans. Fruitless, fifty years out, was still puzzling the best of the science community – could further study actually bring us closer to understanding divergent behavior between the sexes?

Though fruitless retained mystery, the fly field did not rest. Just how did FruM impart aggression in male flies? Researchers at Caltech, led by David Anderson, had spent over a decade developing automated tools for monitoring a variety of fly behaviors, so they undertook a modern version of Gill’s original observational study: manipulate fly genetics systematically, and document the consequences for fly behavior. Other groups had shown the importance of particular hormones, also known as neuropeptides, in fruitless-mediated courtship, which led Anderson’s group to speculate that an unknown neuropeptide could be responsible for male fly aggression.
Harnessing genetic tools for screening many mutant flies at once, the Anderson group created forty fly lines in which small groups of neurons could be activated by heat, depending on which one of twenty neuropeptides they expressed, hypothesizing that some of these lines would display increased aggression. Of these forty lines, two stood out as having increased aggression (for flies, this includes stereotyped lunging and boxing behavior). Both of these lines contained neurons that produced the neuropeptide tachykinin (Tk), also known as Substance P in mammals – a promising early observation for connecting aggression across species. When the researchers obtained high-quality images of these neurons within the fly brain, they noticed that they visually resembled FruM neurons identified in other studies. Digging deeper, lo and behold, this handful of neurons actually expressed FruM, corroborating the earlier work of Dickson’s lab, but refining the aggression-responsible group from two thousand FruM neurons to just four Tk/FruM neurons.
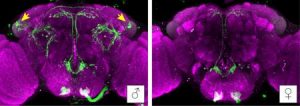
The hunt was on. Anderson’s team characterized how Tk neurons sparked aggression, and found that overexpressing Tk itself, while simultaneously activating the Tk neurons, ramped up aggressive behavior to the point of driving flies to attack inanimate objects (in this case, a fly-sized magnet). Despite also expressing FruM, activating these neurons had no effect on courtship, indicating their specific role in increasing ‘aggressive arousal,’ or the likelihood of engaging in aggressive behavior. These neurons were only observed in males, as expected for FruM neurons, potentially explaining female flies’ low levels of observed aggression. And perhaps most intriguingly, the neuropeptide Tk itself was driving aggression, presenting the first molecular link between aggression in flies and mammals.
With a gene and a neuropeptide pinned down as the factors responsible for male aggression in flies, the Anderson lab, and indeed the entire field studying aggression, now has a new lead on the genetic basis of gender differences in aggression. Will we soon understand why men and women demonstrate different levels and kinds of aggressive behavior? Maybe not for some time. But a similar, aggression-related molecule is present in the mammalian brain, and the fruit fly’s behavior provides the foundation for further study into how sexual dimorphism occurs in higher organisms.The fruitless gene, over forty years since its inception as fruity, continues to bear intellectual fruit, as the grandfather of behavior-regulating genes known to be expressed in a sex-based fashion. Good science takes time, some fumbles with inappropriate names, and some gambles, but the modest fruit fly has yet again proven it is not to be underestimated. Especially when it is up for a fight.
[…] article is being cross-listed on the PLOS Student Blog. Check out some other really interesting pieces […]