Sperm: A ‘Coming of Age’ Journey
This year in the United States, almost 7 million people will encounter the problem of infertility and will likely require some form of assistance or fertility treatment. Many potential parents will opt for in vitro fertilization treatment, a process by which mature eggs is removed from the ovaries and fertilized outside the body in the lab. After fertilization, the formed zygote is implanted into the uterus of the woman whose eggs were used in the treatment, or a surrogate. Since 2003, in vitro fertilization treatments in the United States have risen by 65 percent. This trend is due in part to more people choosing to postpone having children. Research shows parental age affects fertility and the chance of conception, and is associated with unexpected pregnancy complications. In 45 to 50 percent of cases, infertility can be attributed, solely or partially, to the male partner. Although in some cases, the cause of infertility is diagnosable, for example urogenital abnormalities, 40 percent of cases are idiopathic, meaning there is no identifiable cause for observed sperm abnormalities and infertility. Though the underlying cause of infertility is unknown in these cases there are some factors that have been suggested. One probable factor is abnormalities during sperm maturation, particularly at the protein level.
Why Sperm?
Spermatozoa, the mature male sex cells in sexually reproducing species, are among the most rapidly diversifying cell types currently known – showing an awe-inspiring array of morphological and molecular diversity. The morphological diversity of spermatozoa is likely due to intense sexual selection acting on sperm within the female reproductive tract. Sexual selection is defined as a mode of selection acting on traits influencing an individual’s competitive ability to obtain mates and fertilizations. Although sexual selection is traditionally considered a process that occurs before mating, an example being males competing for the attention of females or displaying/posturing to potential mates, sexual selection also takes place in the female after mating is complete as sperm compete to fertilize available eggs. Despite the array of morphological diversity, all sperm function in the same capacity — aiming to deliver the paternal genome to the egg and form a viable zygote. Sperm known best for this essential function. A sperm cell and egg each contain half the genetic material required to form a new offspring. When the two meet, fertilization takes place and a viable zygote is formed. Fertilization thus depends upon the sperm and egg physically meeting, which is the responsibility of sperm; the smaller and more motile of the two. In reproduction, therefore, sperm act as a vehicle to deliver the father’s genetic contribution to the egg, without which fertilization could not take place.
The Molecular Biology of Sperm
Scientific understanding of the molecular biology of sperm has increased considerably in the past decade due, in no small part, to the development of proteomic technology. Proteomic techniques are used to accurately catalog the proteins contained in a sample (the proteome), which in this case is sperm. Protein synthesis in sperm is thought to only occur during spermatogenesis in the testes, ending before sperm become mature. The absence of protein synthesis relates to the fact that there is no gene expression in mature sperm, thus gene expression analyses are not applicable. Research analyzing sperm proteomes across different species consistently finds a set of rapidly evolving sperm proteins (these are often the proteins acting in sperm-egg interactions). The rapid evolution of some sperm proteins reflects the diversification seen at the morphological level and the action of intense sexual selection. On the flip side, the essential function of sperm in reproduction likely constrains rapid evolution, because any mutation or variation negatively affecting sperm’s ability to fertilize would be strongly selected against. Accordingly, a fraction of sperm proteins are present in the sperm of very different species, for example 20 percent of fruit fly sperm proteins have related genes in the mouse sperm proteome.
These shared proteins are part of a ‘core sperm proteome’ involved in necessary metabolic pathways or structural sperm elements, and thus may be essential to sperm function and fertility. In support of this, proteomic studies comparing the proteome of normal/fertile and abnormal/infertile sperm have highlighted many sperm proteins related to fertility. These proteins may be used as markers for fertility screening, or as targets for male contraceptives. Clearly, the sperm proteome can reveal a lot about the molecular composition of sperm, and its analysis has wider reaching implications in health and fertility.
The Journey to Maturation
In vertebrates, sperm exit the testes as immature gametes, immotile and incapable of fertilization. During transit through the epididymis, the sperm gains motility and the ability to fertilize. The epididymis is a highly convoluted tubule running from the testes to the vas deferens (the duct that conveys sperm to the urethra), measuring one meter in mice and three to six meters in humans. It is generally split into three gross regions, the caput (head) , corpus (body) and cauda (tail) , in order from the testes to the vas deferens (see figure 1). Each region maintains an individual microenvironment on which sperm maturation is dependent. As mentioned previously, protein manufacture in sperm halts before exiting the testes, thus the maturing process is dependent on exogenous proteins supplied by the epididymis.
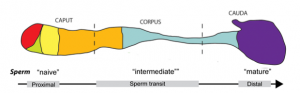
Epididymal maturation is essential for the production of fertile and motile sperm making it an important aspect to consider when assessing male infertility. Furthermore, there is evidence suggesting gene expression in the epididymides is altered in infertile men compared to fertile men.
Combining Proteomics and Sperm Maturation
Considering the utility of proteomics in studying sperm biology, and the importance of sperm changes in the epididymis, applying proteomics to the different epididymal regions seems like a good first step in understanding sperm maturation. However, isolating and purifying sufficient sperm from the three regions is easier said than done and, because it requires dissection of the male reproductive system, is ethically impractical in humans. A recently published paper in PLOS ONE by Skerget and colleagues has, nevertheless, accomplished just this using a mouse model. Researchers applied proteomic analyses to sperm taken from the three main regions of the epididymis to track changes in the protein composition of maturing sperm. By tracking changes to the sperm proteome, they are able to confirm that sperm maturation is a dynamic process at the protein level, involving both the addition and the removal of proteins. The paper focuses on these added/removed proteins, characterizing their predicted functions. The function of a protein is predicted by similarity to proteins, or protein regions, with known and experimentally tested functions. The authors find that a higher than expected proportion of proteins added to sperm during transit are involved in sperm capacitation (the penultimate step in sperm maturation occurring in the female), motility and sperm-egg recognition. Proteins removed from sperm appear more likely to be involved in sperm remodeling, which, presumably, are not required by the mature spermatozoa once remodeling is complete. This finding supports the hypothesis that fertilizing ability and motility is acquired during epididymal transit.
However, the strength of this paper lies not in characterizing the gross changes through the epididymis, but rather the region-specific dynamics of sperm maturation. The authors observed clear distinctions between regions in terms of protein function, with membrane and membrane organization proteins more enriched in the caput, followed by structural proteins in the corpus and lastly proteins relating to sperm function in the cauda. These distinct differences between regions proposes a ‘conveyor belt’ of trackable sperm modifications through the epididymis, with particular changes occurring sequentially in specific regions. The authors identified 60 proteins known to lead to abnormal sperm when mutated or absent, making this study particularly relevant to studies of infertility and sperm maturation. Interestingly, one third of these proteins were among those added to sperm during transit through the epididymis, suggesting the importance of the epididymis in fertility.
Although the findings of the PLOS ONE paper take a step towards understanding sperm maturation in the different regions of the epididymis, there is some way to go before we truly understand the dynamics of a sperm cell’s journey to maturity. Once sperm maturation is understood further factors contributing to idiopathic male infertility can be identified, in particular proteins necessary for correct sperm maturation. These proteins could both be targets for fertility treatment or male contraceptives.
The recent societal shift towards postponing raising children, and the corresponding increase in infertility problems and fertility treatments, makes understanding the factors underlying male fertility increasingly relevant, particularly assessing the links between fertility and aging.
References
Ashrafzadeh, A., Karsani, S.A., Nathan, S. (2013). Mammalian sperm fertility related proteins. International Journal of Medical Sciences. 10(12):1649-57. doi: 10.7150/ijms.6395
Cornwall, G. A. (2009). New insights into epididymal biology and function. Human Reproduction Update, 15(2):213–227. doi.org/10.1093/humupd/dmn055
Dorus, S., Wasbrough, E., Wilkin, E., Hester, S., Howard-Murkin, J., Lilley, K. and Karr, T.L. (2010) The Drosophila melanogaster sperm proteome-II (DmSP-II). Journal of Proteomics 73: 2171-2185. Special Issue on Model Organism Proteomics
Dorus, S., Parker, E.R., Busby, J., and Karr, T.L. (2010) Sperm Proteomics Reveals Intensified Selection on Mouse Sperm Membrane and Acrosome Genes. Molecular Biology and Evolution 27(6):1235–1246
Dubé, E., Hermo, L., Chan, P.T., Cyr, D.G. (2008). Alterations in gene expression in the caput epididymides of nonobstructive azoospermic men. Biology of Reproduction. 78(2):342-51
Jungwirth, A., Diemer, T., Dohle, G.R., Giwercman, A., Kopa, Z., Krauz, C., and Tournaye, H. (2015). Guidelines on Male Infertility. European Association of Urology.
Jungwirth, A., Giwercman, A., Tournaye, H., Diemer, T., Kopa, Z., Dohle, G., and Krausz, C. (2012).
